Amgen’s history is a definitive narrative of the biotechnology industry’s evolution, transitioning from a venture-backed startup in Thousand Oaks to a global biopharmaceutical titan. Here is the breakdown of Amgen’s history by phase:
1. The Founding and Scientific Exploration (1980–1983)
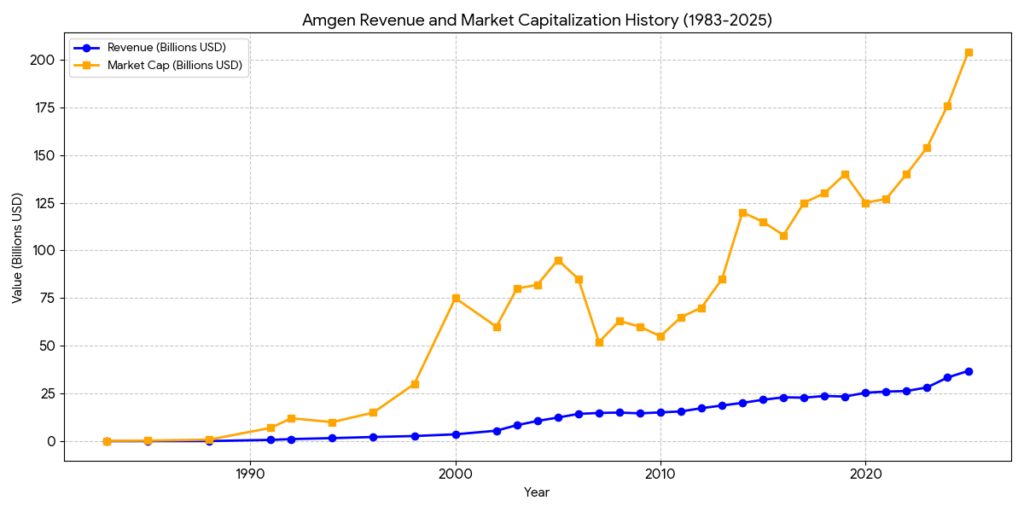
Amgen was founded on April 8, 1980, as Applied Molecular Genetics (AMGen).
- Key Figures: Founded by venture capitalist William Bowes and led by George Rathmann (former Abbott VP), who is often cited as a visionary in the biotech sector.
- Early R&D: The company initially explored a wide array of genetic applications, including extracting oil from shale, rapid-growth chickens, and creating indigo dye for blue jeans using recombinant DNA.
- The Turning Point: In 1983, the company shortened its name to Amgen and went public on NASDAQ. That same year, Dr. Fu-Kuen Lin successfully cloned the erythropoietin (EPO) gene, which would later become the most successful drug in biotech history at that time.
2. The Era of Blockbuster Breakthroughs (1989–1998)
This phase established Amgen as a commercial powerhouse through the launch of two massive “blockbuster” drugs.
- Epogen (1989): The FDA approved recombinant human erythropoietin for treating anemia associated with chronic renal failure. It became the first biotech drug to reach over $1 billion in annual sales.
- Neupogen (1991): Approved for treating chemotherapy-induced neutropenia (low white blood cell count). This cemented Amgen’s dominance in supportive oncology care.
- National Recognition: In 1994, Amgen received the National Medal of Technology for its contributions to the medical field.
3. Diversification and Strategic M&A (2001–2011)
Facing potential patent expirations and a need for growth, Amgen expanded into immunology and bone health through major acquisitions.
- Immunex Acquisition (2002): Amgen acquired Immunex for $16 billion, the largest biotech deal at the time. This gave Amgen Enbrel, a leading therapy for rheumatoid arthritis and psoriasis.
- Bone Health Entry: In 2010, the FDA approved Prolia (denosumab) for osteoporosis, which became a critical long-term growth driver.
- Product Evolution: Launched second-generation products like Aranesp (long-acting EPO) and Neulasta (long-acting Neupogen) to maintain market share.
4. Biosimilars and Cardiovascular Expansion (2012–2019)
Amgen pivoted to address the rising cost of healthcare by developing biosimilars while still pushing for high-end innovation.
- Biosimilar Strategy (2012): Amgen launched an ambitious program to develop biosimilars for competitors’ drugs (e.g., Mvasi, Kanjinti), leveraging its expertise in complex biologic manufacturing.
- Repatha (2015): The launch of Repatha (a PCSK9 inhibitor) marked Amgen’s major entry into the cardiovascular market for lowering LDL cholesterol.
- Asian Expansion: In 2019, Amgen acquired a 20.5% stake in BeiGene to significantly expand its footprint in the Chinese market.
5. Rare Diseases and Precision Medicine (2020–Present)
The current phase focuses on high-margin, specialized medicine and aggressive portfolio transformation.
- Horizon Therapeutics Acquisition (2023): Amgen completed its largest acquisition to date ($27.8 billion), gaining Tepezza (for thyroid eye disease) and pivoting heavily into the rare disease sector.
- Precision Oncology: In 2021, Amgen received accelerated approval for Lumakras, the first-ever treatment for KRAS G12C-mutated lung cancer, solving a target once thought “undruggable.”
- Current Standing: As of 2026, Amgen is a member of the Dow Jones Industrial Average with revenues exceeding $35 billion, focusing on oncology, inflammation, bone health, and rare diseases.
Here is the competitive analysis for Prolia (Denosumab), featuring a comparison of clinical efficacy and safety data with key competitors and their respective manufacturers.
1. Clinical Efficacy Comparison: BMD & Fracture Risk
Prolia is an Antiresorptive agent, while its primary high-end competitors like Evenity and Forteo are Anabolic agents designed to build new bone.
| Comparison Item | Prolia (Denosumab) | Evenity (Romosozumab) | Forteo (Teriparatide) | Bisphosphonates (e.g., Fosamax) |
| Company | Amgen | Amgen / UCB | Eli Lilly | Merck / Various |
| Mechanism | RANKL Inhibitor | Sclerostin Inhibitor | PTH Analog | Bone resorption inhibitor |
| Lumbar Spine BMD Increase | Sustained increase over 10 years (~21%) | Rapid increase in 12 months (~13%) | Significant increase over 18-24 months | Slower increase with a plateau |
| Vertebral Fracture Reduction | ~68% reduction | ~73% reduction | ~65% reduction | ~40-50% reduction |
| Non-vertebral Fracture | ~20% reduction | Superior to Prolia alone | Superior to Bisphosphonates | Moderate effect |
| Dosing Frequency | Subcutaneous every 6 months | Monthly SC (Limited to 12 months) | Daily SC injection | Daily Oral or Yearly IV |
2. Safety Data Comparison
Safety profiles are a major differentiator for Prolia, especially regarding specific patient populations.
- Boxed Warnings:
- Prolia (Amgen): Carries a warning for Advanced Chronic Kidney Disease (CKD) patients due to the high risk of severe hypocalcemia.
- Evenity (Amgen/UCB): Carries a warning for Cardiovascular Risk; it is not recommended for patients who have had a myocardial infarction or stroke within the past year.
- Long-term Side Effects (ONJ & AFF):
- Both Prolia and Bisphosphonates are associated with rare occurrences of Osteonecrosis of the Jaw (ONJ) and Atypical Femoral Fractures (AFF) with long-term use.
- Evenity and Forteo treatments are typically limited to 1–2 years, resulting in a lower cumulative risk of these specific long-term events.
- Rebound Effect (The “Pro-Rebound”):
- Prolia’s Unique Risk: Discontinuing Prolia without transitioning to another therapy causes a rapid decline in BMD and a sharp increase in the risk of multiple vertebral fractures.
- Forteo (Eli Lilly) also sees BMD loss after cessation, but the rebound effect is generally less aggressive than that of Prolia.
3. Market Competitive Position (2026 Outlook)
- Sequential Therapy Trend: The clinical paradigm is shifting toward “Anabolic First, Antiresorptive Second.” High-risk patients often start with Evenity (Amgen/UCB) for 12 months to build bone rapidly, followed by Prolia (Amgen) for long-term maintenance.
- The Biosimilar Threat: Amgen is preparing for competition from Sandoz and other manufacturers developing Denosumab biosimilars. This is expected to erode Prolia’s premium pricing, potentially making it a first-line replacement for traditional oral bisphosphonates in moderate-risk patients.
- Adherence Advantage: Prolia’s bi-annual dosing remains its strongest competitive moat, offering significantly higher patient compliance compared to Forteo’s daily injections.
The following is the competitive analysis of Enbrel (Etanercept), updated with 2025 financial data and 2026 clinical landscape comparisons.
1. Clinical Efficacy Comparison: ACR & PASI Data
As a TNF Alpha Inhibitor, Enbrel remains a foundational therapy, but it is increasingly outperformed by JAK Inhibitors (e.g., Rinvoq) in rheumatoid arthritis (RA) and IL-23 Inhibitors (e.g., Skyrizi) in plaque psoriasis (PsO).
| Product | Enbrel (Etanercept) | Humira (Adalimumab) | Rinvoq (Upadacitinib) | Skyrizi (Risankizumab) | Stelara (Ustekinumab) |
| Manufacturer | Amgen | AbbVie | AbbVie | AbbVie | J&J |
| Mechanism | TNF Inhibitor | TNF Inhibitor | JAK Inhibitor | IL-23 Inhibitor | IL-12/23 Inhibitor |
| RA (ACR50) | ~40-45% | ~45-50% | ~60-70% (Superior) | N/A | N/A |
| PsA (ACR50) | ~35-40% | ~40-45% | ~50-60% | ~50-55% | ~35-40% |
| PsO (PASI 90) | ~45-50% | ~50-55% | ~70-75% | >85% (Superior) | ~55-60% |
| Dosing | Weekly SC | Every 2 Weeks SC | Daily Oral | Every 3 Months SC | Every 3 Months SC |
- RA Leadership: Rinvoq has demonstrated superiority over Enbrel in multiple head-to-head trials, particularly for patients who failed to respond to initial methotrexate therapy.
- PsO Dominance: Skyrizi has set a new efficacy bar in psoriasis, with most patients achieving near-complete skin clearance (PASI 90/100) compared to Enbrel’s more modest PASI 75 baseline.
2. Safety Data Comparison
Enbrel’s primary competitive advantage is its 25-year safety track record, making it a preferred choice for conservative physicians or specialized populations (e.g., pediatric JIA).
- Enbrel (Amgen):
- Side Effects: Injection site reactions, upper respiratory infections.
- Warnings: Serious infections, tuberculosis reactivation, and rare cases of lymphoma.
- Advantage: Known safety profile for long-term use; less metabolic interference than JAK inhibitors.
- Rinvoq (AbbVie):
- Boxed Warnings: Carries FDA warnings for MACE (Major Adverse Cardiovascular Events), blood clots, cancer, and increased mortality risk, limiting its use in older patients or those with high cardiovascular risk.
- Skyrizi (AbbVie):
- Safety Profile: Generally considered to have the most favorable safety profile among biologics, with low infection rates and no Boxed Warnings for heart issues or cancer.
3. Market and Financial Status (2025-2026)
- Revenue Decline: In 2025, Enbrel’s sales experienced significant pressure. In Q3 2025, sales dropped 30% year-over-year to $580 million, primarily due to lower net selling prices caused by the Medicare Part D redesign and increased commercial discounts.
- The “Patent Thicket”: Amgen successfully defended its patents in the U.S., delaying biosimilar competition for Enbrel until 2029. However, indirect competition from Humira biosimilars (launched in 2023-2024) has significantly compressed pricing across the entire TNF class.
- Strategic Shift: Amgen is increasingly focusing on Tezspire (Asthma) and MariTide (Obesity) to replace the revenue lost from the aging Enbrel franchise.
Summary
In 2026, Enbrel is transitioning from a “first-line powerhouse” to a “mature stable asset.” While it remains a multi-billion dollar product, its growth has peaked. Its role is now focused on patients who prioritize long-term safety data or who prefer a proven biologic over the newer, more potent but potentially higher-risk JAK inhibitors.
Source:
- https://www.amgen.com/newsroom/press-releases/2026/02/amgen-reports-fourth-quarter-and-full-year-2025-financial-results
- https://investors.amgen.com/news-releases/news-release-details/amgen-reports-third-quarter-2025-financial-results/
- https://www.amgen.com/about/amgen-history
- https://www.alpha-sense.com/earnings/amgn/
- https://www.investing.com/news/transcripts/earnings-call-transcript-amgen-q3-2025-results-exceed-expectations-stock-rises-93CH-4332439
- https://pmc.ncbi.nlm.nih.gov/articles/PMC8528978/
- https://www.buzzrx.com/blog/evenity-vs-prolia
- https://en.wikipedia.org/wiki/Amgen
- https://swottemplate.com/blogs/brief-history/amgen-brief-history
Back to Amgen page
